26 June 2023. New restrictions for several cosmetic substances, including Retinol, will enter into force in the European Union by the end of 2023. On June 8, 2023, the European Commission notified the World Trade Organisation (WTO) of a draft regulation changing the regulatory status of certain ingredients.
The draft introduces the following changes to the EU Cosmetics Regulation:
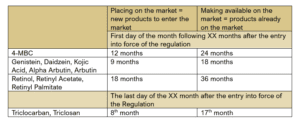
- Prohibition of 4-Methylbenzylidene Camphor (4-MBC) and consequent deletion of the relevant entry in Annex VI (list of UV filters);
- Restrictions for Genistein (up to 0,007%), Daidzein (up to 0.02%), Kojic Acid (only in face and hand products up to 1%), Alpha-Arbutin (up to 2% in face cream and 0,5% in body lotion), Arbutin (up to 7% in face cream), and Vitamin A;
- Further limitations in Annex V for the preservatives Triclocarban and Triclosan:
- Triclocarban will be allowed in all cosmetic products, except for mouthwash, up to a maximum concentration of 0,2%;
- Triclosan will be banned in mouthwash;
- Additionally, toothpaste with Triclocarban or Triclosan will have to bear the warning “Not to be used for children under 6 years of age” as those substances cannot be used in toothpaste for children under 6.
New restrictions for Vitamin A
Vitamin A and its derivatives are used in several skin care products as skin conditioning agents. As Vitamin A naturally occurs in fish, eggs, dairy products, and others, humans are exposed to it not only from cosmetics but also from other sources, mostly from food and food supplements.
In its opinion of October 2022, the SCCS confirmed that the overall exposure of consumers to Vitamin A might exceed the upper limit determined by the European Food Safety Authority (EFSA), although cosmetics contribution is lower than other sources.
Furthermore, the SCCS identified maximum safe limits for the use of Vitamin A in cosmetic products. Consequently, the European Commission transposed them into the new draft regulation. The restrictions concern the following forms of Vitamin A: Retinol, Retinyl Acetate, and Retinyl Palmitate.
According to the draft Regulation, those substances will be allowed for use up to a maximum concentration of 0,05% of Retinol Equivalent (RE) in body lotion and up to 0,3% RE in other cosmetics. The labelling of those products will also have to include the statement “Contains Vitamin A-related compounds, which contribute to your daily intake of Vitamin A”.
Deadlines for the application
The European Commission plans to publish the regulation in the fourth quarter of 2023 officially. The draft sets some deadlines for applying the new restrictions. In this way, the industry has time to adapt, avoiding excessive financial and environmental costs. The details are in the table below.
If you are a beauty brand, you shall check the formulation of your products and take the necessary measures to ensure compliance considering the above-mentioned new restrictions. Acting already at this stage shows your company’s commitment towards safety and will ensure you will be ready by the deadlines.
Become a COSlaw member for free to enjoy our exclusive content, or contact us at coslaw@obelis.net for more information.
References:
- European Commission. (2023). Commission Regulation (EU) …/… of XXX amending Regulation (EC) No 1223/2009 of the European Parliament and of the Council as regards the use of Vitamin A, Alpha-Arbutin and Arbutin and certain substances with potential endocrine disrupting properties in cosmetic products. Retrieved on 19/06/2023
- European Commission. (2023). Annex to the Commission Regulation …/… Retrieved on 19/06/2023
- WTO. (2023). Notification G/TBT/N/EU986. Retrieved on 19/06/2023
- SCCS. (2022). Revision of the scientific opinion (SCCS/1576/16) on vitamin A (Retinol, Retinyl Acetate, Retinyl Palmitate). Retrieved on 19/06/2023



Leave a Reply